 The electronic structures of the d block elements shown are: Sc The first row of these is shown in the shortened form of the Periodic Table below. The elements in the Periodic Table which correspond to the d levels filling are called d block elements. At argon, the 3s and 3p levels are full, but rather than fill up the 3d levels next, the 4s level fills instead to give potassium and then calcium. You will remember that when you are building the Periodic Table and working out where to put the electrons using the Aufbau Principle, something odd happens after argon. They don't - there's a subtle difference between the two terms. The terms transition metal (or element) and d block element are sometimes used as if they mean the same thing. They are generally very stable chemically and exhibit similar properties of being colorless and odorless.\) They generally very chemically reactive and are present in the environment as compounds rather than as pure elements. They comprise group 17 of the periodic table, from F through At. The halogen elements are a subset of the nonmetals. The term "nonmetals" is used to classify the elements H, C, N, P, O, S, and Se. Only thorium and uranium are naturally occurring actinides with significant abundance. They, along with the lanthanides, are often called "the f-elements" because they have valence electrons in the f shell. The actinides comprise elements 89 through 103. They, along with the actinides, are often called "the f-elements" because they have valence electrons in the f shell. They are grouped together because they have similar chemical properties. The lanthanides comprise elements 57 (lanthanum, hence the name of the set) through 71. 
They sometimes behave as semiconductors (B, Si, Ge) rather than as conductors. The metalloids are B, Si, Ge, As, Sb, Te, and Po. Metalloid (or "semi-metal" or "poor metal").They tend to be softer and conduct more poorly than the transition metals. As their name implies, they have some of the characteristics of the transition elements. The post-transition elements are Al, Ga, In, Tl, Sn, Pb and Bi. The transition elements are metals that have a partially filled d subshell (CRC Handbook of Chemistry and Physics) and comprise groups 3 through 12 and the lanthanides and actinides (see below). Their characteristics are well described and consistent down the group. The alkaline earth metals have very high melting points and oxides that have basic alkaline solutions. The alkaline earth metals make up group 2 of the periodic table, from Be through Ra. Hydrogen is group 1 but exhibits few characteristics of a metal and is often categorized with the nonmetals. They have very similar behavior and characteristics. 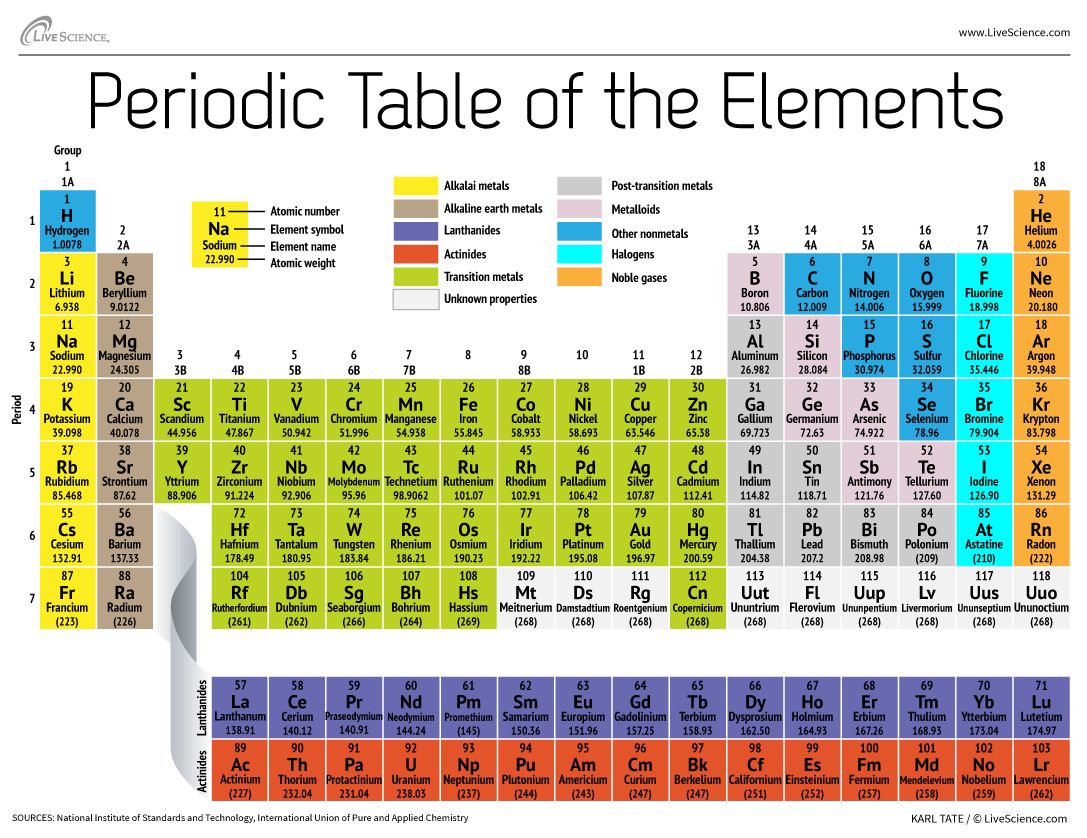
The alkali metals make up group 1 of the Table, and comprise Li through Fr. In our version of the table, we have chosen the most commonly accepted demarcations between these elements. For example, in some tables, Group 12 is is categorized with the post-transition metals, and in others, aluminum and tin are included characterized as Metalloids or poor metals. The dividing line between metals and non-metals is not hard and fast, thus the distinction between "Post-transition metals" and "Metalloids" is represented differently on different versions of the Periodic Table. Metal elements are usually good conductors of both electricity and heat. 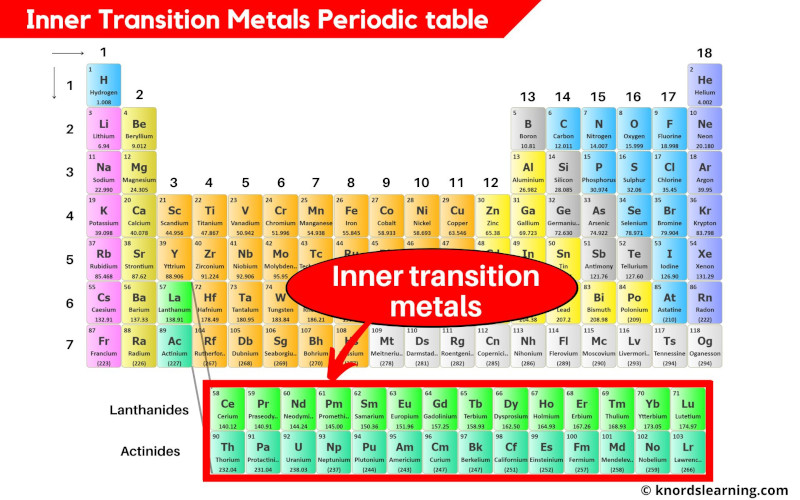
Elements can generally be described as either metals or nonmetals. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
